
We compared coated surfaces and the benefits of moisture management to understand how to know when a coating is hydrophobic. Here's what we learned.
How Do I Know I Have a Water Repelling Coating?
|
In this blog post you will learn:
- How surface energy changes moisture resistance and hydrophobicity
- The benefits of a hydrophobic coating surface
- Compare the water repelling properties of silicon coatings
|
A hydrophobic surface is a water repelling, low surface energy surface that resists wetting. Moisture contact angle measurements will classify a surface as hydrophobic when the contact angle of the water droplet exceeds 90 degrees. Exceed the 150 degree contact angle mark and the surface will be classified as super hydrophobic. Water will jump right off the surface as seen in this video.
How to tell if a surface is water repelling. Measuring hydrophobicity.
The contribution of hydrophobicity, water repelling surface energy and it's relationship to process fluids can have far ranging impacts to products and processes. Surface interaction can impact corrosion, fouling, analytical sampling results, filtration and medical device performance. So it's important to understand how surface energy relates to hydrophobicity and to measure and manage the surface energy of coatings or surfaces.
Have a question about this blog or moisture resistance? Click the box below to discuss your application with one of our coating scientists.
Have a Question? Contact Our Technical Service Team
How to measure water repelling properties on a surface
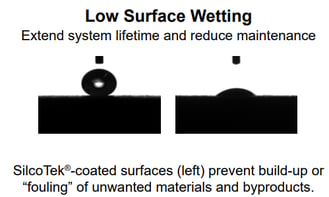
The analysis of interaction of a liquid with a surface can provide a simple but powerful understanding of the surface’s characteristics and how it will perform in other related environments. Water droplet contact angle data using liquids of known surface tensions can provide an insightful characterization of a surface that has been tailored to exhibit desirable functions. A droplet that remains round when sitting on a surface (above left image) has a high water droplet contact angle and is said to be hydrophobic or water repelling. Compared to the droplet on the left image which lies flat and is wetting the surface (demonstrating a low water droplet contact angle).
A high contact angle with water translates to a hydrophobic surface that may display rapid dry down properties for tubing subjected to gas streams contaminated with water vapor. This may in turn prevent localized corrosion as the water vapor is less likely to pool and corrode the tubing over time.
Contact Angle Measurement

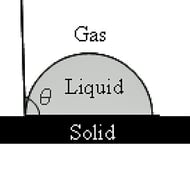
SilcoTek has two instruments for contact angle analysis: a Krüss K100 tensiometer and a Ramé-Hart Model 200 goniometer shown above. Both systems provide valuable information when evaluating the interactive forces between a known liquid and a test surface. The tensiometer is excellent at providing bulk analysis of a larger surface area, generating advancing and receding contact angle values. The goniometer is a fixed level stage system (i.e. no tilt) that can provide a localized equilibrium contact angle. It is also excellent at providing a visual identification of the solid/liquid/air interaction.
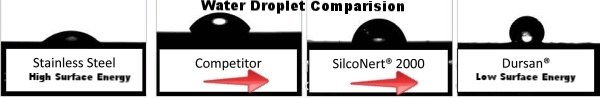
Comparative Test Data
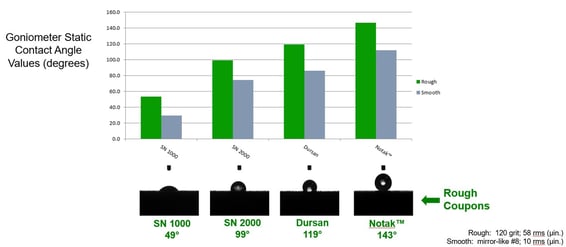
We compared the hydrophobicity of several coated surfaces. The test substrate was 316L stainless steel in two different surface finishes: a corrosion coupon with a 120 grit finish, and a sample coupon with a “mirror finish” surface. Since surface topology can have an effect on the equilibrium contact angle data (measured with the goniometer), a wide variance of roughness was used to help bracket the performance expectations of each surface. The same variance
in surface roughness was used in the tensiometer measurements, but the nature of that measurement
(bulk dip vs. localized drop) is not overtly sensitive to those variations.
The test results below compares the contact angle of several coatings. A basic silicon surface has about the same contact angle as stainless steel. Various coatings shown can drastically change the hydrophobicity of the surface. The lower the surface energy of the coating, the higher the contact angle.
How do I get a surface to match my desired moisture level of moisture resistance?
You don't have to make a super radical change to material or product construction in order to change the moisture performance. Fortunately we offer silicon barrier coatings with a wide range of water management capabilities. Want a more corrosion resistant or inert coating with a low surface contact angle? Try SilcoNert® 1000 or Silcolloy®. If you're looking for an inert coating that's not too moisture repelling, go with SilcoNert® 2000. Need maximum water repelling coating properties? Dursan® or our new Notak coating may do the trick. Note, our Notak coating is in pre-production Beta testing so we have limited capacity for that coating. Contact our Technical Service Team to discuss your application and we'll be happy to make a coating recommendation.
Hydrophobicity Benefits
There are many benefits of a water repelling surface. They include:
| Surface icing prevention |
Condenser & evaporator fouling prevention |
| Improved corrosion resistance |
Prevent moisture contamination in heat trace tubing |
| Prevent metal filter corrosion |
Improved reliability in continuous emissions monitors (CEMS) |
| Improved moisture detection instrumentation |
HPLC medical diagnostics improved separation & corrosion resistance |
| Manage moisture contamination in liquid natural gas systems |
Prevent contamination in analytical sample transfer systems |
Hydrophilic Surfaces
Not to be outdone, hydrophilic surfaces have benefits as well. What are hydrophilic surfaces? They are high surface energy substrates that attract water and allow wetting of the surface. They typically have a droplet contact angle measurement of less than 90 degrees. Lots of surfaces tend to be more water friendly including, glass, steel, or stainless steel and many coatings and paints. Of course test results can depend on the surface roughness and surface energy of the material you're testing.
Learn How To Improve Moisture Resistance, Fouling Resistance, and Corrosion Resistance. Get Our Presentation.
Benefits of a moisture friendly surface include.
| Improved separation in medical diagnostics (application dependent) |
Improved efficiency in heat transfer devices & heat exchangers |
| Improve acceptance of In-body devices |
Improve surface interaction in filtration devices (application dependent) |
Go to our applications guide to get general information about each of our coatings. Or go to our applications web page.

Want to learn more about managing surface energy for improved hydrophobicity and fouling resistance? Watch our webinar.









