
Nearly half of all sample transfer systems have failed or are failing. That can't be good for process control. Get tips on how to keep sample systems running, avoid regulatory penalties, and save money!
Many process control systems require either sample collection or direct interaction with process fluids or calibration systems. That's why the durability, surface reactivity and responsiveness of process sampling and control flow paths are key to plant performance.
|
In this blog post you will learn:
- How to improve the durability and reliability of process control and process sampling systems.
- How to improve the responsiveness of process monitoring systems.
- How chemical vapor deposition can be used to apply high durability non-reactive coatings to existing component designs.
- Key factors to consider when specifying materials used in process control systems and process sampling systems.
|
Challenges facing instrument probes and sampling systems.
Process monitoring systems face many environmental and process challenges, they can range from probe or tube fouling, sample phase change (condensation, freezing, etc.), extreme temperatures, surface reactivity and adsorption, pooling of liquid in the line, moisture contamination, corrosion and the list goes on! That's why there are lots of systems that are in critical condition and require frequent maintenance or replacement.
Get our e-book: 7 Tips For Improved Analytical Sampling for in-depth information on how to keep your analytical system running at peak performance. Or get our latest e-book: The Ultimate Guide to Reliable Analytical Sampling.

Robust system design, a solid maintenance plan, and proper material selection will improve performance and assure regulatory compliance and efficient process control. Working with specialists in heat trace installation will help to avoid common design errors sample transfer problems; while optimum system material selection will require input from in house process engineers, technicians and sample system design engineers; the people who know the plant conditions the best. After assessing the key factors impacting the sampling and control system, it's time to assess material requirements.
Selecting the right material for the application.
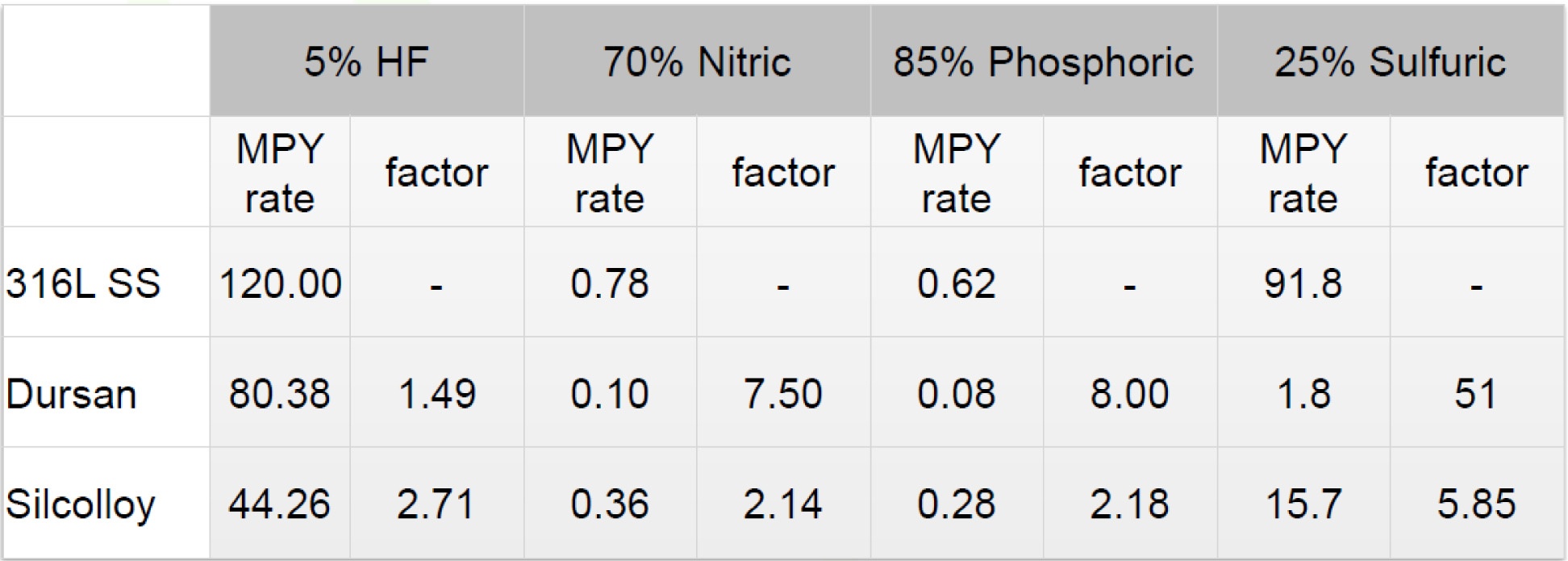
Moving a sample from point A to Point B requires some form of conveyance be it a sample cylinder carried by your feet to the GC or via tubing connected to a continuous emissions monitoring system (CEMS). No matter the transport method, the material a sample comes in contact with during that trip matters. The go-to materials for sample transport are PTFE and stainless steel, but before reaching for those options, you need to consider the sample environment, the nature of the sample being taken, the desired result and cost. This is clearly a one size does not fit all situation. Let's consider a chemical process plant with monitoring probes exposed to several corrosives. The table below shows how stainless steel and CVD coatings perform when exposed to various corrosives. The table shows that depending on the chemical exposure and concentration, corrosion performance can vary significantly. This can directly impact plant performance and maintenance cost.
Environmental considerations
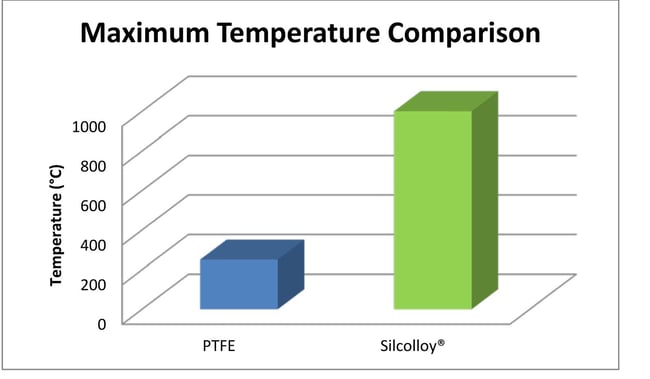
Conditions to consider range from the temperature of the sample environment, exposure to corrosives, potential for damage or abrasion, potential for cold shock or rapid temperature swings. Some sample systems must maintain a steady temperature that comes very close to the maximum temperature of PTFE (250°C). Even if temperatures are relatively cool, say 120°C, PTFE can become soft and deform or "cold flow", resulting in collapse of liners or tubing failure. Extreme cold can embrittle PTFE and flow path liners which can also lead to failures. Super inert coatings like SilcoNert® and Dursan® can withstand high temperature exposure up to 450°C while silicon coatings like Silcolloy® remain intact at temperatures over 1000°C.
Corrosion resistance
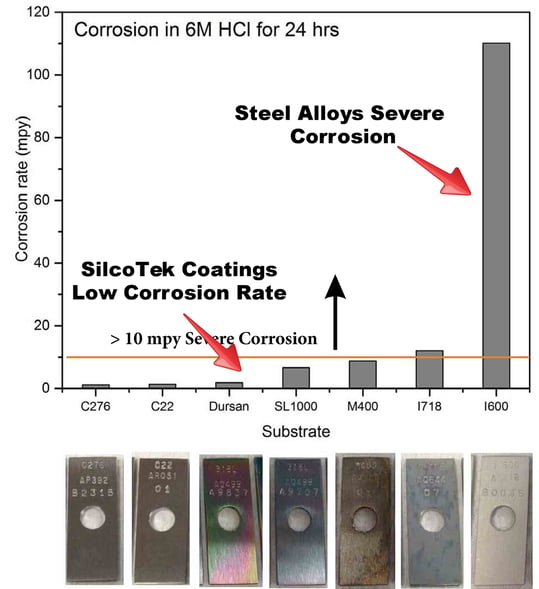
Stack and flare samples often contain acids such as sulfuric acid or hydrochloric acid. Even low level exposure can pit tube or sample cylinder surfaces and eventually lead to system failure. Pitted surfaces can also be great places for trace amounts of moisture and analyte to hide. This can result in sample carryover or moisture solvation of the sample. Inert coatings can improve corrosion resistance of stainless steel, helping to achieve an affordable, high durability, corrosion resistant and inert surface. Comparative analysis of immersion studies of stainless steel, alloys, and SilcoTek® coated stainless steel coupons in 6M hydrochloric acid show an order of magnitude decrease in corrosion for the Dursan coated surface.
Abrasion and wear resistance
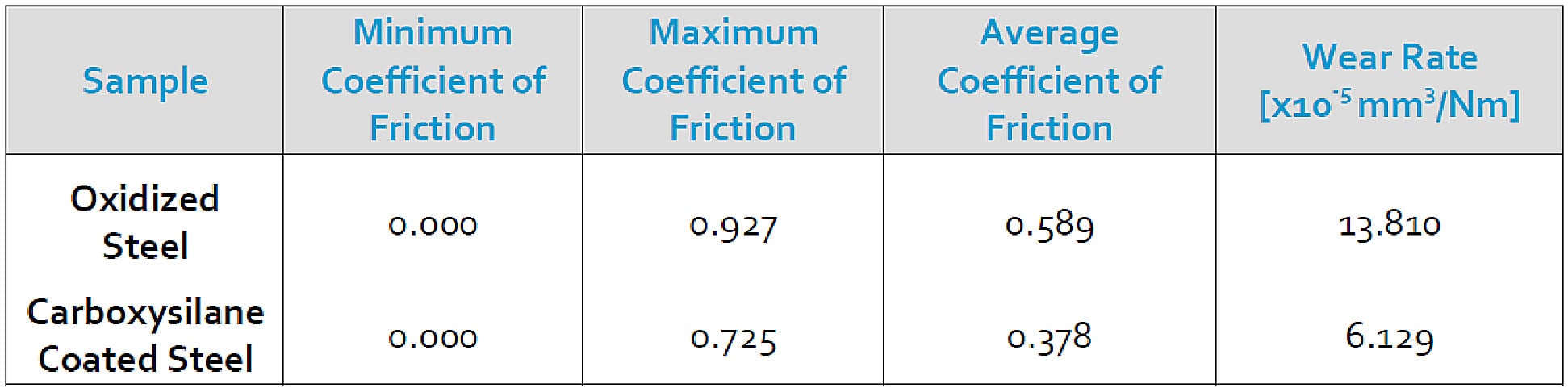
Abrasive samples can quickly damage PTFE and other soft surfaces. Filtration of the sample can help protect the tube or sample cylinder surface but designers should be mindful of potential damage caused by abrasion or simple rough handling by installers and technicians. Pin-on-disc studies comparing Dursan (carboxysilane) coated steel with uncoated steel shows a 50% reduction in wear rate and a significant reduction in friction, which can improve durability and efficiency of sample systems. Dursan, and all our CVD coatings can be applied to internal and external surfaces. Our process is non line-of-sight so we're able to coat very small diameter tubing and even sintered metal fritted filters.
Watch our video and see how abrasion can damage flow path surfaces.
Know the sample
It sounds obvious, you're sampling for whatever is specified say VOC's or H2S. That's all I need to know right? Sorry, you'll need to know lots more including:
- Sample phase (don't change from gas to liquid during sampling, or visa versa...)
- Corrosive compounds in the sample
- Moisture content
- Sample temperature and pressure
- Can potential compounds in the sample react with the flow path materials, including seals?
Know the expected concentration range of the sample when sampling in analytical applications. For example a 20% H2S sample will need a more robust flow path vs. a 20 ppb sample. Conversely, surface reactivity will have a greater relative impact on the 20ppb sample. A 10 ppb loss due to flow path adsorption results in a 50% loss to the trace sample but a negligible impact to the 20% sample.
Achieving desired results
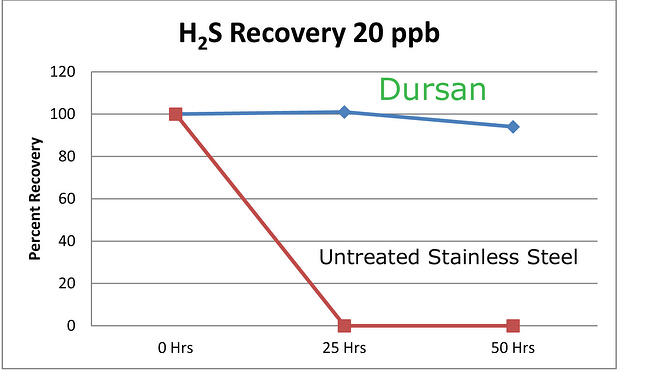
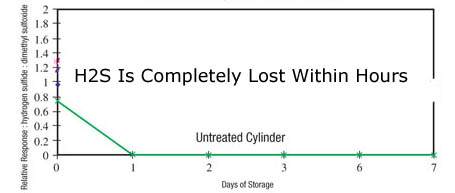
Are you taking a quick grab sample for a maximum allowable value? Are you calibrating the system for ultra low level analysis? Or are you speciating a sulfur sample? Knowing where you're going with the sample system performance is key to the material selection and ultimate system performance. Conversely, don't expect a sample system designed for sampling 20% H2S to magically be capable of accurately and reliably sampling H2S at 20ppb. Hint, you'll never see the 20ppb sample. In fact reactive samples can be adsorbed by stainless steel, alloy, glass, ceramic or elastomer surfaces on contact. Often a trace sample when held in an untreated sample cylinder will be completely lost due to adsorption.
How to improve response
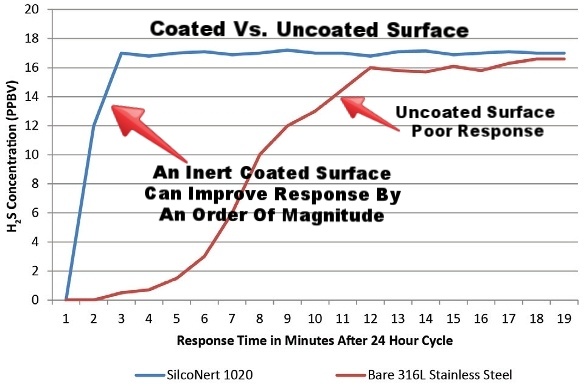
That adsorption due to surface interaction can be a real problem. Not only can you not get accurate results, the whole sample system can spiral out of control. That's because adsorption and reactive surfaces can slow response time and can slow the sample from getting to your analyzer or sample system. Here's an example, say you've got a hydrogen sulfide (H2S) sample from a stack sampling CEMS. As the sample travels through a reactive sample line, some of the H2S can attach or adsorb to the sample line surface. As the sample travels through foot after foot of sample line pretty much all the sample is lost by the time it hits the sample instrument. Only after all the active sites are full of H2S will the subsequent sample pass. How long can that take? Minutes to hours depending on the length of line and amount of H2S in the sample. The coated line, which is non adsorptive, allows the sample to race through the line and hit the analyzer unmolested, cutting the response time to seconds rather than minutes or hours. This can be a real asset when product yield or regulatory compliance depends on response and process control.
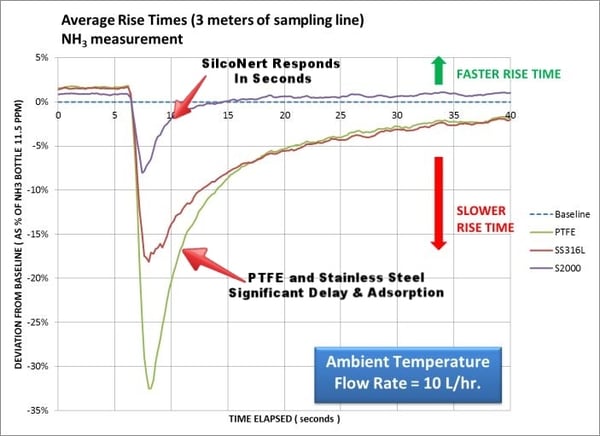
Fast response is not only limited to H2S or sulfur compounds. Pretty much any reactive sample can benefit from a Dursan or SilcoNert coated sample line. Here ammonia is injected into 3 meters of sampling line. The SilcoNert coated line responds in seconds while the stainless steel and PTFE line takes significantly longer to reach the baseline response. Delays in response can cause false alarms, cause auto feed systems to improperly meter product or can damage catalyst or process systems.
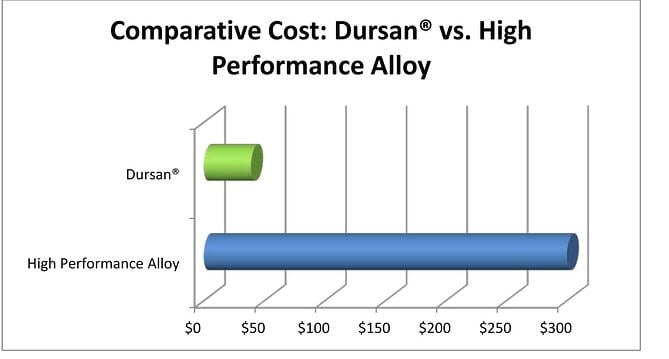
Cost
We all have unlimited budgets so spend, spend, spend! OK maybe not, so cost will be a factor. Not just the initial cost, but the total operating cost of the sample system over the life of the system or project. A quick calculation will prove that frequent repairs to a system will quickly eat up any potential short term savings realized by not taking into the noted system design factors, or by simply going with stainless steel rather than an inert system.
Material reference
Confused about choosing the best material for an analytical flow path? Here's a quick comparison of material advantages and limitations for sample transfer applications. Or you can contact our Technical Service Team and we'll be happy to discuss your application.
| Material |
Advantages |
Limitations |
| Stainless Steel |
Low cost, durable, high/low temperature capable |
Reactive, lower corrosion resistance |
| PTFE |
Corrosion resistant, inert, low cost |
Permeable, easily damaged, low melting point |
| Super Alloys |
Corrosion resistant |
Expensive, limited availability, reactive surface |
| Inert Coating, Dursan, SilcoNert |
Corrosion resistant, inert, durable, temperature capable |
Can be damaged by base exposure. Abrasives can damage over time. |
Want more tips on improved surface inertness and better analytical results?













