
A CVD coating is a material applied to a surface by a method called chemical vapor deposition. Let's discuss how the coatings are made, coating benefits, and how to get the most out of your coating.
CVD coatings are prevalent throughout industry and can be found in many consumer products. They're known as being an environmentally friendly durable thin film surface. CVD products can be found in applications ranging from machine tools, wear components, analytical flow path components, instrumentation and many other areas demanding a high performance thin film. There are lots of thin films out there so what is it about the chemical deposition process that makes a superior coating for high performance precision applications?
Have a question about this blog or CVD coatings or coating selection? Click the box below to discuss your application with one of our coating scientists.

|
In this blog post you will learn:
- How CVD (chemical vapor deposition) works.
- Characteristics of a CVD process
- Benefits and drawbacks of chemical vapor deposition.
- Why our coatings are so colorful.
|
About CVD Coatings: What is Chemical Vapor Deposition?
Chemical vapor deposition is a process that involves the reaction of a volatile precursor which is injected into a chamber (typically under vacuum). The chamber is heated to a reaction temperature that causes the precursor gas to react or break down into the desired coating and bond to the material surface. Over time the coating material builds on the surface and creates a coating throughout the exposed part's surface. So for example if we wanted to bond silicon to a surface we may want to use a trichlorosilane precursor. When the trichlorosilane is heated in the coating chamber the decomposition and coating reaction may look like this.
SiHCl3 → Si + Cl2 + HCl
The silicon will bond to any exposed surfaces (both internal and external) while the chlorine and hydrochloric acid gas will be vented from the chamber and scrubbed according to appropriate regulatory requirements. The CVD coating system may look something like the diagram below. In this example the desired coating material is a silicon oxide base coating with a functionalized (R) surface which is intended to cap any remaining active sites on the coating surface. This makes for a superior non reactive surface. Want to learn more about our coating process? Go to our Coating Technology and Quality page.
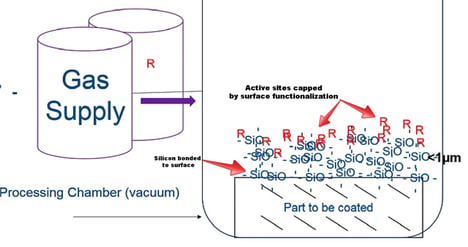
Materials used in CVD coating systems range from silicon compounds to carbon, to fluorocarbons or organofluorine, and nitrides like titanium nitride. Some materials like silicon can be further enhanced by doping the surface with other materials to functionalize the surface for a specific performance goal (as noted above).
Need help selecting the right CVD coating for your application? Try our online coating selector guide.

Selecting the right coating for the application is important, but coating quality control, (including surface preparation, process control, coating precursor, and material substrate quality) play a major role in the coating quality. The info graphic below shows a typical CVD coating process.
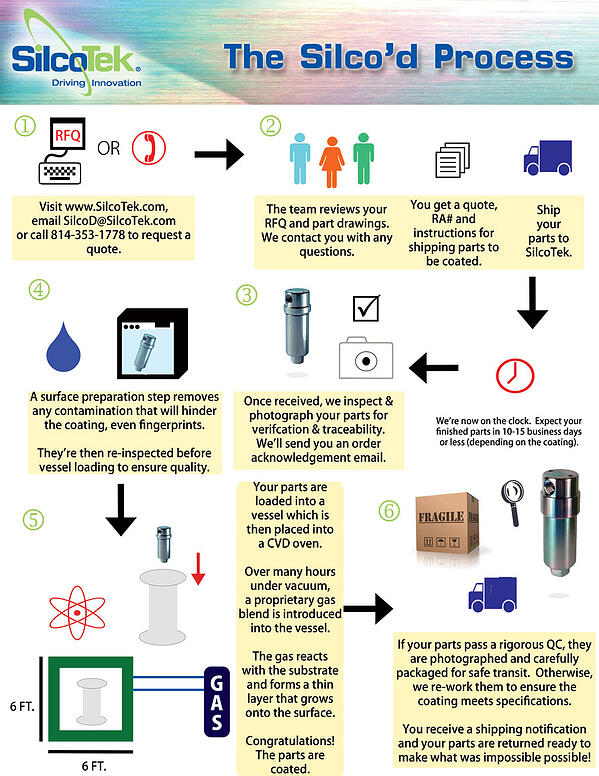
Characteristics of a CVD coating process
- Applied at elevated temperature to facilitate reaction.
- Typically applied under vacuum.
- Contaminants must be removed from the part surface before coating application.
- Process may limit the base materials that can be coated. I.e. temperature limitations or reactivity limitations.
- Process may limit the ability to mask specific target areas.
- Unlike most PVD (physical vapor deposition) processes, the CVD Process is not limited to line-of-site application. Coating gas will coat all areas of a part including threads, blind holes, and interior surface.
- Film thickness is limited due to coating stress.
- Coating is bonded to the surface during the reaction which creates a superior adhesion when compared to typical PVD or low temperature applied spray coatings.

Coating Benefits
- Can be applied to a wide variety of base materials including ceramics, glass, metals and metal alloys.
- Can coat precision surfaces and intricate surfaces including seal areas and internal surfaces
- Can withstand exposure to low and high temperature and extreme temperature variation
- Remains bonded in high stress environments and when the surface flexes due to high adhesion characteristics.
- Precursor gas can be optimized for wear resistance, high lubricity, corrosion resistance, fouling resistance, high purity, or chemical inertness.
Have a question about a coating application? Get a free technical consultation with one of our coating experts.

Coating Drawbacks
- Typically applied at higher temperatures (depending on the precursor).
- Difficult to mask surface. Usually an all or nothing coating.
- Size limited to reaction chamber capacity.
- Parts must be broken down into individual components and is not an "on site" process.

The CVD coating process and how it makes color.
Customers often make note of the multi-color rainbow appearance of our coatings. What makes the coated part so colorful and why does the color vary by part or lot?
We bond silicon to metals, glass, or ceramics, this process allows us to build a silicon layer on a surface one Angstrom (0.1 nanometer) at a time. A super thin layer of silicon is pretty clear, in fact you can see through a thin silicon layer. As the silicon thickness increases on say a stainless steel surface, light which travels through the silicon is bent. The light then reflects off the stainless surface and is bent again. All this light bending causes some wavelengths of light to cancel out each other while other wavelengths are reinforced. The canceled (or interference) wavelengths aren't seen by your eye while the reinforced wavelengths hit your cones and are seen as color. Changing the color of a thin silicon surface is easy, a few nanometer change in silicon thickness can have a big impact on the colors you see.
 The best analogy of this effect that I can relate to is oil on water. I'm sure you've noticed that after a rain storm a colorful rainbow oil slick can be found flowing from under that 1966 Mustang that's been parked in your driveway and waiting to be restored for the past 8 years. I know you'll get to it.... Anyway, oil is not colorful, it's either honey colored when new or black when draining from your oil pan. That black oil is becoming a colorful rainbow because of refraction. Light reflects off the top of the oil surface and also travels through the thin oil and is reflected back from the water surface below the oil. Depending on the thickness of the oil, light rays will either be reinforced or interfered with by the oil refraction. When the rays of light reach your eye, the destructive interference wavelengths cancel each other out. The oil thickness variation dictates what colors you see and what color wavelengths get canceled out. So a slight variation in thickness can have a big impact on the color you see. *
The best analogy of this effect that I can relate to is oil on water. I'm sure you've noticed that after a rain storm a colorful rainbow oil slick can be found flowing from under that 1966 Mustang that's been parked in your driveway and waiting to be restored for the past 8 years. I know you'll get to it.... Anyway, oil is not colorful, it's either honey colored when new or black when draining from your oil pan. That black oil is becoming a colorful rainbow because of refraction. Light reflects off the top of the oil surface and also travels through the thin oil and is reflected back from the water surface below the oil. Depending on the thickness of the oil, light rays will either be reinforced or interfered with by the oil refraction. When the rays of light reach your eye, the destructive interference wavelengths cancel each other out. The oil thickness variation dictates what colors you see and what color wavelengths get canceled out. So a slight variation in thickness can have a big impact on the color you see. *
The same holds true with silicon. Small variations in thickness can make a big difference in the color of the coating. So the different colors observed on Silco'd treated parts indicate different layer thicknesses. A blue color corresponds to a 300 to 450 Angstrom layer while a rainbow color indicates a coating of at least 1200 Angstroms (120 nanometers).
Colors associated with layer thickness are:
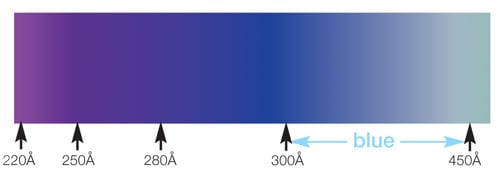
Depositions used in our Silcolloy® and SilcoGuard® processes are up to 1µm (1000nm) and have a rainbow to silver/metallic gray appearance. The photos below show colors created by SilcoNert®, a 500nm** coating (left) and Silcolloy®, an 800nm** coating (right). So as you can see, a few nanometers change in thickness will result in a dramatic change in the color of the part.

Our coatings are colorful because they're thin. Why do we make the coating thin? Because the amorphous silicon coating when thin does not impact part tolerance significantly and because our coatings are highly durable so they don't need to be thick.
Have other questions? Try our Frequently Asked Questions page.

So why don't we make parts all the same color? We're an industrial coating company so we're focused on the ultimate performance of the coating, not appearance. We could in theory make parts with uniform color, but that would impact coating thickness and may result in a less robust coating. Plus rainbow is more fun!
*Image credit: https://www.pinterest.com/explore/oil-spill/








 The best analogy of this effect that I can relate to is oil on water. I'm sure you've noticed that after a rain storm a colorful rainbow oil slick can be found flowing from under that 1966 Mustang that's been parked in your driveway and waiting to be restored for the past 8 years. I know you'll get to it.... Anyway, oil is not colorful, it's either honey colored when new or black when draining from your oil pan. That black oil is becoming a colorful rainbow because of refraction. Light reflects off the top of the oil surface and also travels through the thin oil and is reflected back from the water surface below the oil. Depending on the thickness of the oil, light rays will either be reinforced or interfered with by the oil refraction. When the rays of light reach your eye, the destructive interference wavelengths cancel each other out. The oil thickness variation dictates what colors you see and what color wavelengths get canceled out. So a slight variation in thickness can have a big impact on the color you see. *
The best analogy of this effect that I can relate to is oil on water. I'm sure you've noticed that after a rain storm a colorful rainbow oil slick can be found flowing from under that 1966 Mustang that's been parked in your driveway and waiting to be restored for the past 8 years. I know you'll get to it.... Anyway, oil is not colorful, it's either honey colored when new or black when draining from your oil pan. That black oil is becoming a colorful rainbow because of refraction. Light reflects off the top of the oil surface and also travels through the thin oil and is reflected back from the water surface below the oil. Depending on the thickness of the oil, light rays will either be reinforced or interfered with by the oil refraction. When the rays of light reach your eye, the destructive interference wavelengths cancel each other out. The oil thickness variation dictates what colors you see and what color wavelengths get canceled out. So a slight variation in thickness can have a big impact on the color you see. * 


